Complete Guide on Electrochemical Machining
Categories
Introduction
Electrochemical Machining (short for ECM as below) has become a valuable choice for manufacturing in-demand machined and engineering parts. Various machining processes are used worldwide to machine different metals to give them a desirable shape and surface finish. Electrochemical machining involves slow production and achieves impressive precision to make ECM more prominent than other methods.
W.Gusset developed this technique in 1929, which involves the removal of material from metal through electrochemical means.

What is Electrochemical Machining?
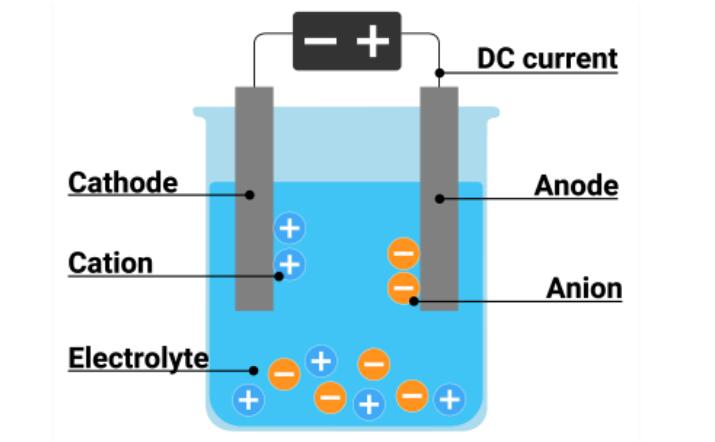
It is a machining process involving removing material from a workpiece through an electrochemical process. It includes a cathode, anode, and electrolyte, commonly NaCl. During the process, the tool acts as a cathode while the workpiece acts as an anode.
Both these electrodes are allowed to be submerged in electrolytes and are placed closely without touching each other. After immersing in the electrolytes, the voltage is applied across the electrodes. It results in the removal of material from the workpiece. It produces a mirror finish surface because of the removal of material at the atomic level.
Types of Electrochemical Machining
Electrochemical machining is divided into the following types.
1. Electrolytic Processing
It is an electrochemical reaction that involves dissolving an anode to form metal by using a large operating current.
During the process, the distance between the workpiece anode and the tool cathode remains unequal because of the continuous repelling of the tool towards the workpiece. The distance between both electrodes becomes smaller when the tool comes close to the workpiece. At this point, the maximum dissolving of the workpiece occurs because of the highest density of current. The electrolyte removes the electrolysis product, and the desired shape of the anode is attained.
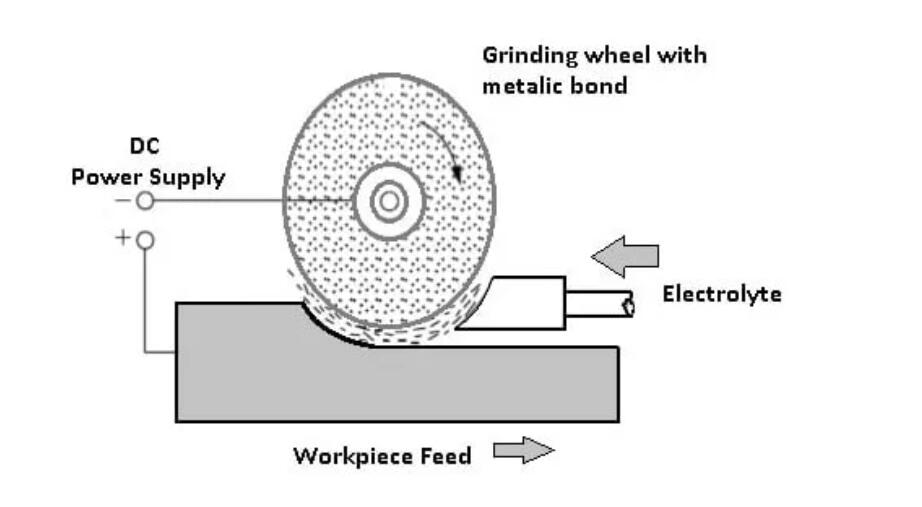
2. Conductive Grinding
Conductive grinding, also called electrolytic grinding, combines mechanical grinding and electrolysis.
During the process, the conductive grinding wheel is connected with the cathode of the DC power supply. On the other hand, the workpiece is connected to the anode, and the electrolyte is added to the processing zone. After turning on the power, the anodic dissolving of the surface of the metal of the workpiece takes place. It results in the formation of a very thin layer of oxide on the metal surface. The grinding wheel did the polishing of the oxide layer.
3. Electrochemical Polishing
Electrochemical polishing, also termed electrolytic polishing, is a process in which the direct processing of metal parts improves the surface finish of the metal by dissolving the anode through an electrochemical reaction.
Metal hardness does not affect the whole process. Its principle is similar to electrolytic machining. The main difference is that the cathode is fixed during the process. It causes a larger gap between the poles and a small amount of metal removal. A controlled current density is essential during electropolishing. A large current density reduces current efficiency, while a small current density results in low production efficiency and the metal's corroded surface.

4. Electroplating
It is a process that involves the deposition of a metal on another conductor or non-conductor surface through electrolysis. It enhances metals' electrical conductivity, wears resistance, and corrosion resistance properties.
During the process, the metal to be plated is connected to the anode while the other metal is connected to the cathode. The ions present in the electrolytic solution are similar to the ions of metal present at the anode. As the process begins, the anode starts dissolving into the metal solution.
At the same time, the equal number of metal ions present in the solution starts gaining the electrons present on the cathode. Then, they keep precipitating on the metal surface that requires plating until a plated metal layer is formed.

5. Electroetching
Electroetching is also called electrolytic etching. It is a process in which the principle of electrochemical anodic dissolution is used to etch the desired or specific text or pattern on the surface of a metal. The processing principle of electroetching is similar to electrolytic machining.
During electroetching, the amount of metal removal is extremely low. Thus, it does not require a high-speed flowing electrolyte to remove the workpiece's dissolving product. Moreover, the fixing of the cathode is done during processing.
6. Electrolytic Smelting
Electrolytic smelting is a process in which the electrolysis principle is used to refine rare and colored metals. It is mainly classified into two types.
- Baking salt electrolysis smelting
- Aqueous electrolytic smelting
Baking salt electrolysis smelting
It extracts and refines active metals such as aluminum, magnesium, sodium, etc.
Aqueous salt electrolysis smelting
The metallurgical industry uses it to extract and refine metals such as nickel, lead, and zinc.
Advantages of Electrochemical Machining
Electrochemical machining offers the following advantages.
- The machining operations show high accuracy resulting in fewer errors. So, it is best to obtain accurate dimensions.
- You can achieve a high-quality metal surface by using this machining process.
- During the machining operations, no burr formation occurs.
- There is negligible production of stresses on the surface of the workpiece during the machining process.
- The machining process does not cause any tool wear.
- The workpiece hardness does not affect the machining rate.
- No heat is produced during the process.
Application of Electrochemical Machining
Major applications of electrochemical machining are listed below:
- It is used for drilling and milling operations and Die-sinking operations. It is also used for the multiple holes drilling process.
- You can machine turbine blades by using this process. The complex concave structure of the turbine blades makes them tough to the machine by other processes.
- You can produce tiny gear systems during this process which are difficult to machine by other machining methods.
- Because of its high surface finish and accuracy, you can use it for micromachining, contouring, and profiling
- Hard metals can be heavily machined with the help of this machining process.
Conclusion
Electrochemical machining applies to the removal of material from a metal part. Horizontal and vertical types of ECM machines can use for this process. Machine type and size depend on their working requirements. The various types of electrochemical machining provide specific surface finishes to the metals.
So, these are widely used to give the desired surface finish to your metal components. Although it is not used for soft materials, its surprising benefits and wider applications make it a dominant process over the other machining processes on an industrial scale.
Worthy Hardware is a CNC manufacturing and sheet metal fabrication company,including CNC machining services,CNC milling services, CNC turning services, laser cutting services and stamping services.Call us +86-76989919645 or email us [email protected] for more discounts for your projects.

